Spatial structure of an example protein containing an iron-sulfur centre
Dr hab. Rafał Dutkiewicz, prof. UG from the Intercollegiate Faculty of Biotechnology UG and MUG, has received funding of PLN 688,080 in the PRELUDIUM BIS 5 competition announced by the National Science Centre.
The funding was awarded in the Life Sciences category for the project The in vivo role of the HscA/HscB chaperone protein system in the biogenesis of iron-sulphur (Fe-S) centres in Escherichia coli. We talk to prof. Rafal Dutkiewicz about the research plans and their breakthrough.
Julia Bereszczyńska: - What is the aim of your research?
Dr hab. Rafał Dutkiewicz, prof. UG: - Currently, our research is focused on understanding the molecular mechanisms of the involvement of the chaperone proteins Hsp70 and cooperating J-proteins in the biogenesis of iron-sulphur (Fe-S) centres, which are prosthetic groups essential for the function of numerous proteins. The biological utility of the centres is due to their unique ability to bind and release electrons. Proteins containing Fe-S centres function in key cellular processes: oxidative phosphorylation, nitrogen assimilation, metabolite synthesis, ribosome biogenesis, DNA replication and repair. Despite their simple structure, the biogenesis of Fe-S centres is a complex and poorly understood process involving specialised proteins that form multiprotein complexes. Protein-protein interactions between components of these complexes are crucial for their activity. To date, our research has focused on the identification and biochemical characterisation of these interactions using purified proteins involved in the biogenesis of Fe-S centres in Escherichia coli.
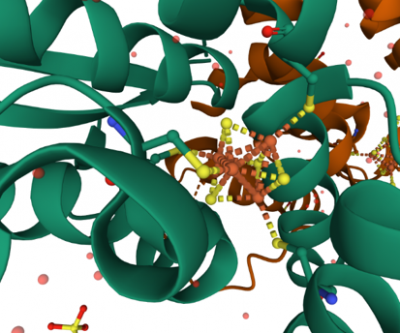
Close-up of the 4Fe-4S centre. The 4Fe-4S centre is coordinated by four cysteine residues. The yellow colour indicates the sulphur atoms and the red colour indicates the iron atoms.
- What is the groundbreaking nature of your research?
- Despite detailed biochemical analysis, little is known about the importance of these interactions for the biogenesis of Fe-S centre-containing proteins in vivo. In this project, we will develop a research model to study the physiological consequences of perturbations of specific interactions between proteins involved in Fe-S centre biogenesis in vivo. To this end, we have established a collaboration with dr Beatrice Py (Aix-Marseille Université-CNRS), who is an expert in the genetic study of the biogenesis process of bacterial Fe-S centres. We will prepare an E. coli strain allowing the expression of genes encoding variants of proteins defective in specific protein-protein interactions, which will allow us to study the role of these interactions during the synthesis of proteins containing Fe-S centres. The hallmark of this project is the innovative combination of detailed biochemical studies with in vivo analyses. This will provide insight into the molecular mechanisms underlying this important metabolic process.
- What impact can perturbations of specific interactions between proteins involved in Fe-S centre biogenesis have on the organism’s functioning?
- The proteins involved in Fe-S biogenesis are evolutionarily conserved and orthologues of the proteins we studied have also been identified in mammals, including humans. This strong conservatism can be explained by the fact that Fe-S centres produced in mitochondria are essential for the survival of every eukaryotic cell. Thanks to this conservatism, experimental results obtained using bacteria or yeast as convenient model organisms are easily translatable to the human system. This is of great importance because in vertebrates, and especially in mammals, deletion of individual genes encoding proteins involved in mitochondrial Fe-S biogenesis is lethal at early stages of embryonic development. In humans, mutations that damage the function of these genes lead to numerous diseases, often resulting in death at a very young age. Conditions associated with disorders of Fe-S biogenesis include Friedrich's ataxia, myopathy with lactic acidosis and various types of syroblastic anaemia.
- Is there a chance that, with this research, it will be possible in the future to treat diseases that are caused by disorders of Fe-S biogenesis?
- Since disorders of Fe-S biogenesis in humans, for example as a result of mutations in the proteins involved, are the cause of many diseases that are often fatal, the results we have obtained will help to understand the molecular basis of these conditions and perhaps, in the long term, contribute to the development of effective therapies.
