
Publication authors
The publication titled 'Human EXOG Possesses Strong AP Hydrolysis Activity: Implication on Mitochondrial DNA Base Excision Repair', authored by scientists from the Intercollegiate Faculty of Biotechnology UG&MUG Faculty of Biotechnology: dr hab. Michał R. Szymański, prof. UG and dr Anna Karłowicz, has been published in the prestigious scientifi Journal of the American Chemical Society (JACS).
Link to publication: https://doi.org/10.1021/jacs.2c10558
Abstract
Mitochondria are essential to the life of the eukaryotic cell due to their key role in essential metabolic pathways. The proximity of the respiratory chain, which is the source of reactive oxygen species (ROS) in mitochondria, makes mitochondrial DNA (mtDNA) much more vulnerable to oxidative stress than nuclear DNA. The discovery that unrepaired damage leads to the destabilisation of the mitochondrial genome, resulting in ageing and many serious diseases in humans, started intensive research into the mechanisms of mtDNA repair. Still, the repair processes in mitochondria are not fully understood and many fundamental questions still need to be answered.
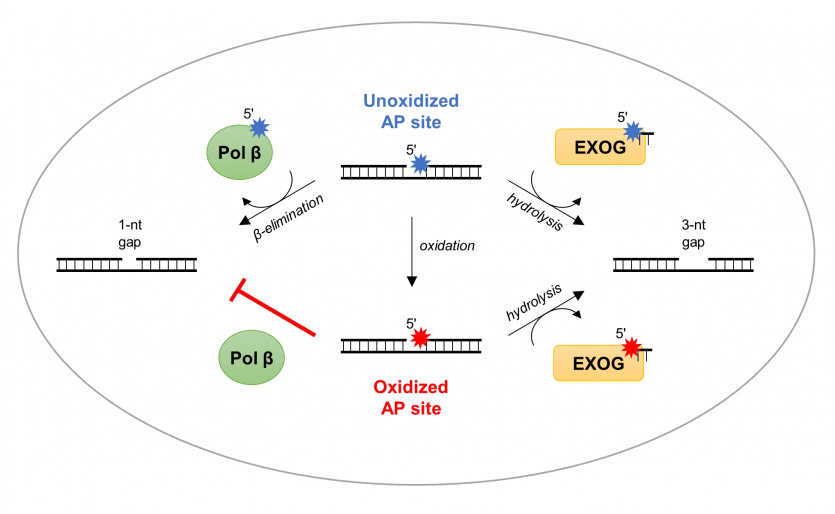
Most oxidative damage in mtDNA is repaired through the Base Excision Repair (BER) pathway, in which many enzymes are involved. However, the mitochondrial enzyme responsible for removing deoxyribose phosphate (dRP) from the 5' end has not yet been described. The 5'-dRP is formed after removal of the damaged base (thus creating an AP site) and incision of the sugar-phosphate backbone of the DNA.
- 'Through structural and enzymatic analyses in our work, we have shown that the human mitochondrial exonuclease EXOG has very strong dRP removal activity. In contrast to canonical dRP lyases (e.g. repair polymerase Pol β in the cell nucleus), which remove only intact dRP, EXOG is capable of excising different AP sites: (1) dRP, (2) deoxyribonolactone dL, an oxidised form of dRP, and (3) tetrahydrofuran THF, a synthetic analogue of dRP. We solved the crystal structure of EXOG in complex with THF-containing DNA, as well as with a partial nucleotide break, explain the authors of the publication. - The structural data obtained, together with biochemical analyses, allowed us to propose a mechanism for EXOG activity on DNA substrates containing different damage. We have shown that, through a controlled 5'-exonucleolytic activity, EXOG incises the third phosphodiester bond from the AP site, allowing the removal of damage in mtDNA regardless of its type,' - they add.
The work was carried out under the POLONEZ 2 grant of the National Science Centre (NCN), of which dr hab. Michał R. Szymański, prof. UG was the head.
